Projects
- Spinal Cord Injury
- Alzheimer's Disease
- Neural Plasticity
The Aging and Alzheimer’s Disease Research Group in the Center for Neural Repair studies degenerative changes in the brain that are a consequence of both normal aging and pathological disorders such as Alzheimer’s disease. We explore the hypothesis that nervous system growth factors, including Nerve Growth Factor (NGF), Brain Derived Neurotrophic Factor (BDNF), and other trophic molecules influence neuronal survival and function at molecular, cellular, electrophysiological and behavioral levels. These programs contribute to our understanding of mechanisms underlying neuronal vulnerability to degeneration, and represent translational programs for developing more effective therapies for neurodegenerative disorders.
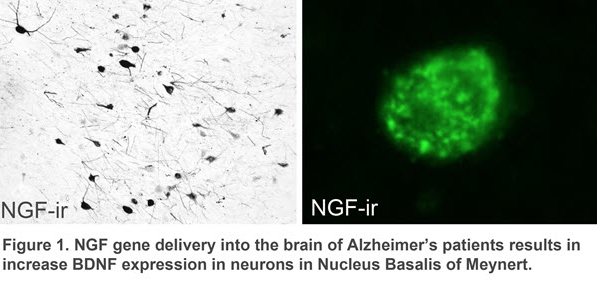
Previous work in the laboratory demonstrated that Nerve Growth Factor (NGF) gene therapy can prevent cholinergic neuronal loss and augment cholinergic function in experimental animal models1. These findings led us to examine the hypothesis that NGF gene therapy would delay or even prevent degeneration of cholinergic neurons in the basal forebrain of patients with Alzheimer’s disease. In 2001, we initiated a program of NGF gene transfer to humans with Alzheimer’s disease, the first human clinical trial of gene therapy in an adult neurological disorder. This work has continued through Phase 2, multicenter human clinical trials. Preliminary results demonstrate that gene therapy results in elevated NGF expression in neurons near the targeted site (Figure 1)2.
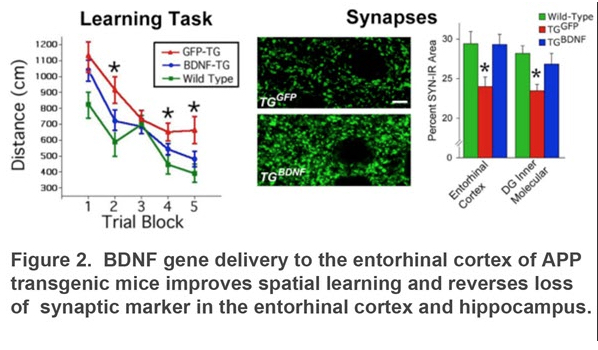
Our current studies focus on Brain-derived Neurotrophic Factor (BDNF). BDNF play a major role in learning, memory, and regulation of synaptic plasticity in the cerebral cortex. In early Alzheimer’s disease, the entorhinal cortex exhibits neuron death that may contribute to loss of memory. We found that BDNF treatment in the entorhinal cortex can improve learning, reduce synapse loss, prevent neurodegeneration, and normalize gene expression in different models of AD (Figure 2)3,4. We are currently working to translate these promising findings to a first-in-human clinical trial using MRI-guided infusion of BDNF gene therapy targeting the entorhinal cortex (Figure 3).
At a more basic level, we are attempting to discover genes that correlate with risk to neurodegeneration as a function of normal aging, and how growth factors modify age-related changes in gene expression. We are further defining how normal aging influences neuronal structure at the level of dendritic spine density and morphology, and the impact of growth factors on these degenerative changes.
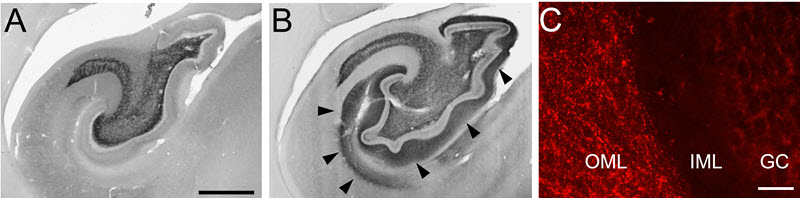
Figure 3. Widespread elevation of BDNF immunolabeling in the hippocampus following AAV2-BDNF delivery to entorhinal cortex of non-human primate. BDNF immunoreactivity in a control subject (A) and treated subject (B); arrow indicate area of elevated BDNF. (C) Fluorescent labeling illustrates BDNF immunoreactive fibers in the outer molecular layer (OML) and not in the inner molecular layer (IML) or granule cell layer (GC) of hippocampus. Scale bar a,b 1 mm; c 200 µm.
1. Castle MJ, Cheng Y, Asokan A & Tuszynski MH (2018). Physical Positioning Dramatically Enhances Brain Transduction After Intrahecal AAV9 Infusion. Sci Adv 4(11): eaau9859.
2. Nagahara AH, Wilson BR, Ivasyk I, Kovacs I, Rawalji S, Bringas JR, Pivirotto PJ, San Sebastian W, Samaranch L, Bankiewicz KS, Tuszynski M. MR-Guided delivery of AAV2-BDNF into the entorhinal cortex of non-human primates. Gene Therapy, 2018, in press.
3. Rafii MS*, Tuszynski MH*, Thomas R, Barba D, Brewer JB, Rissman RR, Siffert J, Aisen PS, and the AAV2-NGF Study Team. A phase II double-blind, placebo-controlled (sham-surgery), multicenter study evaluating AAV2-NGF in vivo gene delivery for Alzheimer’s disease. JAMA Neurology, 2018, in press. *Co-first authors.
4. Wang L, Conner JM, Nagahara AH, Tuszynski MH. Rehabilitation drives enhancement of neuronal structure in functionally relevant neuronal subsets: Cholinergic dependence. Proc Nat Acad Sci, 2016; 115:2750-2755
5. Biane JS, Takashima Y, Conner JM, Massimo Scanziani, Tuszynski MH. Thalamocortical projections exhibit plasticity onto behaviorally-relevant neurons during adult motor learning. Neuron, 2016; 89:1173-1179.
6. Wang L, Conner JM, Nagahara AH, Tuszynski MH. Rehabilitation drives enhancement of neuronal structure in functionally relevant neuronal subsets. Proc Natl Acad Sci, 2016; 113:2750-2755.
7. Tuszynski MH, Yang JH, Barba D, U HS, Bakay R, Pay MM, Masliah E, Conner JM, Kobalka P, Roy S, Nagahara AH. Neuronal responses to nerve growth factor gene therapy in Alzheimer’s disease. JAMA Neurology, 2015; 72:1139-1147.
8. Biane J, Scanziani M, Tuszynski MH, Conner JM. Motor cortex maturation is associated with reductions in recurrent connectivity among functional subpopulations and increases in intrinsic excitability. J Neurosci, 2015; 35: 4719-4728.
9. Ramanathan DS, Conner JM, Anilkumar AA, Tuszynski MH. Cholinergic systems are essential for late-stage maturation and refinement of motor cortical circuits. J Neurophysiol, 2015, 113:1585-1597.
10. Biane J, Conner JM, Tuszynski MH. Nerve growth factor is primarily produced by GABAergic neurons of the adult rat cortex. Front Cell Neurosci, 2014; 8:220.
11. Nagahara AH, Mateling M, Wang L, Eggert S, Rockenstein E, Imracs I, Koo EH, Masliah E, Tuszynski MH. Early BDNF treatment ameliorates cell loss in the entorhinal cortex of APP transgenic mice. J Neurosci, 2013; 33:15596-602
12. Wang L, Conner JM, Rickert JL, Tuszynski MH. Structural plasticity within highly specific neuronal populations identifies a parcellation of motor learning. Proc Nat Acad Sci, 2011, 108:2545-2550. PMCID: PMC3038698.
13. Conner JM, Kulczycki M, Tuszynski MH. Unique contributions of distinct cholinergic projections to motor cortical plasticity and learning. Cerebral Cortex, 2010, 20:2739-2748.
14. Nagahara A, Merrill DA, Coppola G, Tsukada S, Schroder BE, Shaked GM, Wang L, Blesch A, Kim A, Conner JC, Rockenstein E, Chao MV, Koo E, Geschwind D, Masliah, Chiba AA, Tuszynski MH. Neuroprotective effects of BDNF in rodent and primate models of Alzheimer’s disease. Nature Med, 2009, 15:331-337. PMCID: PMC2838375.
15. Ramanathan D, Tuszynski MH, Conner JM. The basal forebrain cholinergic system is required specifically for behaviorally-mediated cortical map plasticity. J Neurosci, 2009, 29:5992-6000.
16. Nagahara A, Merrill DA, Coppola G, Tsukada S, Schroder BE, Shaked GM, Wang L, Blesch A, Kim A, Conner JC, Rockenstein E, Chao MV, Koo E, Geschwind D, Masliah, Chiba AA, Tuszynski MH. Neuroprotective effects of BDNF in rodent and primate models of Alzheimer’s disease. Nature Medicine, 2009, 15:31-337.
17. Conner JM, Franks KM, Titterness AK, Russell K, Merrill DA, Christie BR, Sejnowski TJ, Tuszynski MH. NGF is essential for hippocampal plasticity and learning. J Neurosci, 2009, 29:10883-10889.
18. Ramanathan D, Conner JM, Tuszynski MH. A form of motor cortical plasticity that correlates with recovery of function after brain injury. Proc Natl Acad Sci, 2006; 103 30:11370-11375.
19. Conner JM, Chiba AA, Tuszynski MH. The basal forebrain cholinergic system is essential for cortical plasticity and functional recovery following brain injury. Neuron, 2005, 46:173-9.
20. Tuszynski MH et al. A phase I clinical trial of nerve growth factor gene therapy for Alzheimer’s disease. Nature Med, 2005, 11:551-5.
21. Conner JM, Culberson A, Packowski C, Chiba A, Tuszynski MH. Lesions of the basal forebrain cholinergic system impair task acquisition and abolish cortical plasticity associated with motor skill learning. Neuron, 2003, 38:819-829.
22. Conner JM, Darracq MA, Roberts J, Tuszynski MH. Non-tropic actions of neurotrophins: Subcortical NGF gene delivery reverses age-related degeneration of primate cortical cholinergic innervation. Proc Nat Acad Sci, 2001; 98:1941-1946.